Finally.
Meet FDA Section 524B Cybersecurity Requirements for Medical Devices
Bulletproof Trust supports the evidence, visibility, and workflow controls manufacturers need for FDA Section 524B and premarket cybersecurity documentation — using the same risk data trusted by the FDA to evaluate medical device software.

Proactive FDA Compliance
Reduced Remediation Costs
Secure-by-Design Device Dev
Block Non-Compliance Now
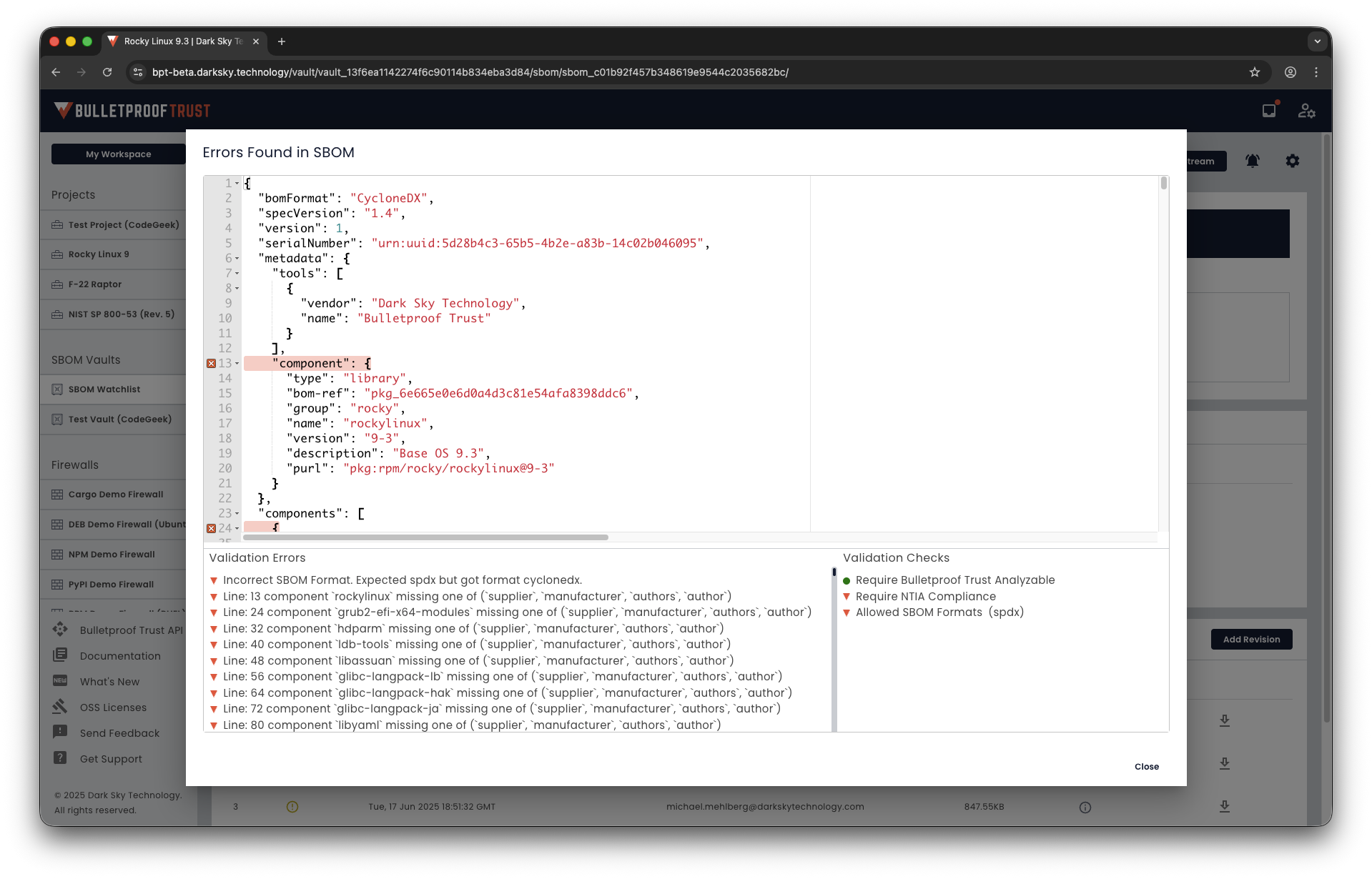
Secure SBOM Storage
Compliance Validation Checks
Audit Traceability
Generate CycloneDX / SPDX

Contributor Trust Intelligence
License Risk Identification
Package Health Scoring
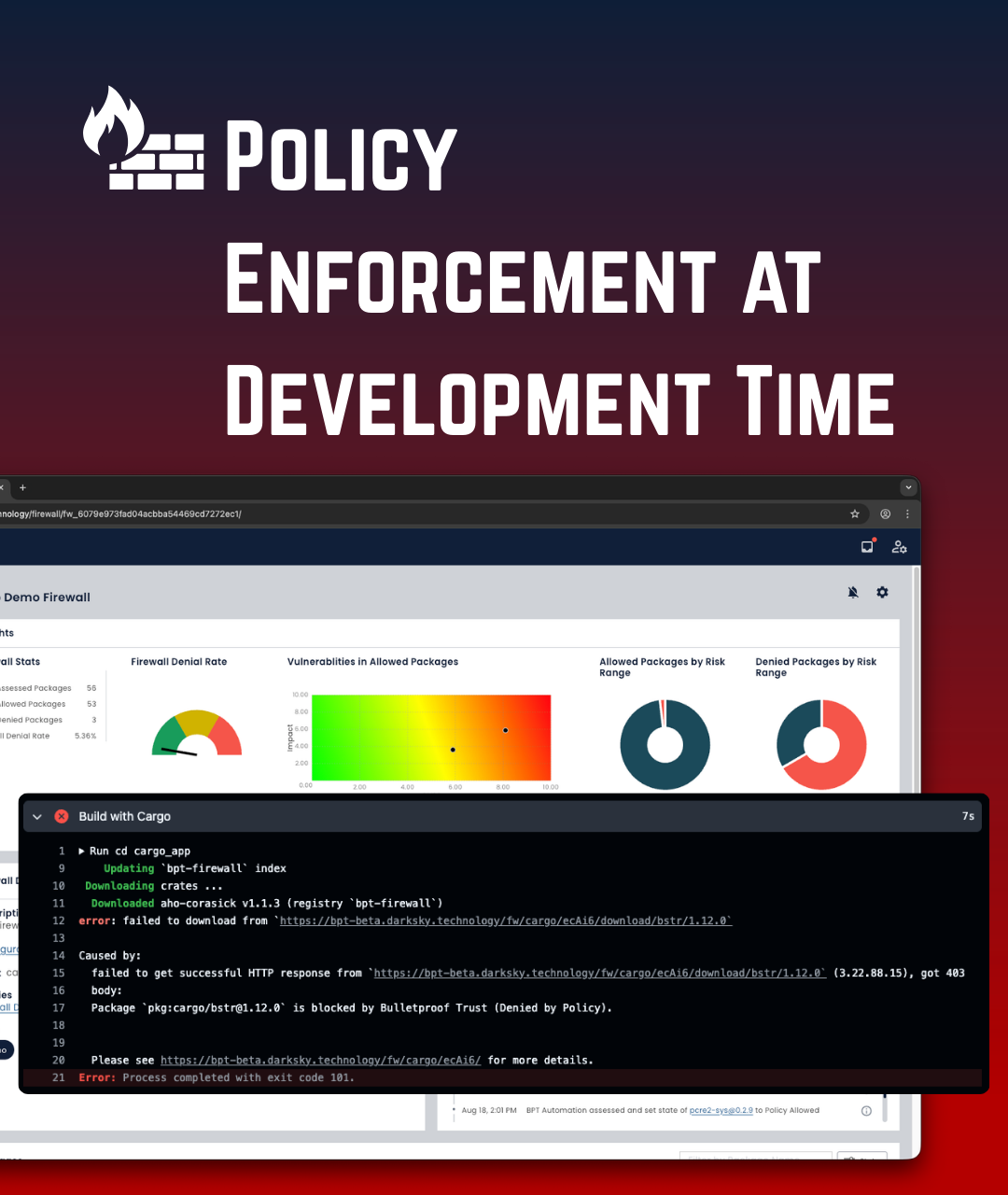
Policy Driven Risk Enforcement

40+ Vulnerability Databases
SBOM Threat Analysis
Automated Risk Comparison
Real-Time Threat Intelligence

The FDA Has Changed the Rules
For devices that meet the FDA definition of a cyber device, Section 524B requires manufacturers to include cybersecurity information in premarket submissions such as 510(k), PMA, De Novo, PDP, and HDE.
Provide an SBOM covering commercial, open-source, and off-the-shelf software
Submit a plan to monitor, identify, and address postmarket cybersecurity vulnerabilities and exploits
Maintain processes and procedures that provide reasonable assurance the device and related systems are cybersecure
Make available postmarket patches and updates to address vulnerabilities
Document cybersecurity controls, risk management decisions, and supporting evidence in premarket submissions
Failure to provide sufficient cybersecurity documentation can result in:
eSTAR technical screening holds
Refuse-to-Accept or acceptance delays
Longer review cycles and additional questions
Late-stage remediation work before clearance or approval
Medical device cybersecurity is no longer best practice — it is regulatory mandate.
The Solution? Bulletproof Trust™
Align your compliance process with the same data source used by the FDA.
Bulletproof Trust is a mature, deployed platform capable of meeting FDA level SBOM scrutiny.
SBOM Transparency
Generate CycloneDX/SPDX SBOMs and manage a complete inventory of third-party and open-source components, complete with version tracking, vulnerability identification, and conformance validation.
Continuous Vulnerability Monitoring
Ongoing monitoring of your entire supply chain across 40+ vulnerability databases. Rapidly assess risk when new CVEs emerge, and document mitigated actions.
Secure Software Development Lifecycle
Aligned to NIST SSDF (800-218) with risk-based decision making and policy-driven controls. Traceable remediation documentation.
Postmarket Risk Management
Continuous SBOM updates and alerting on newly disclosed vulnerabilities. Audit-ready reporting.

Don’t Let Cybersecurity Delay Your FDA Submission
Who is this for? Why Bulletproof Trust?
Who is this for?
Bulletproof Trust is for medical device manufacturers building or maintaining connected products that may qualify as cyber devices, including OEMs, SaMD teams, connected diagnostics platforms, embedded Linux device manufacturers, Class II and Class III device developers, and the regulatory, product security, and engineering teams supporting premarket submissions.
Why Bulletproof Trust?
Bulletproof Trust is a trusted vulnerability and risk information data set for the FDA’s Cybersecurity program that assesses medical device manufacturers cybersecurity compliance. Bulletproof Trust has been deployed on our nations most critical systems, in air-gapped environments, with enterprise teams.
Why Dark Sky Technology?
Dark Sky Technology builds software security products for high-assurance and regulated environments. Bulletproof Trust is deployable in SaaS, on-prem, private cloud, and air-gapped environments for teams that cannot expose sensitive code or development data.
Trusted by the FDA, and deployable in sensitive air-gapped environments.
Built originally to protect critical national-systems, now securing regulated medical technologies.
Designed for Regulated Environments

Your code is sensitive, and you can’t share it with anyone. Bulletproof Trust can deploy in air-gapped environments, so you don’t have to share anything with us.
Deploy Air-Gapped

Personally identifiable information (PII) comes with a whole separate set of rules, regulations, and laws. Bulletproof Trust masks every bit of data so you never have to handle PII.
Avoid Dealing with PII

Connect directly to the API to perform all analysis, manage the platform, and integrate with your build environment. Or, drive Bulletproof Trust from the command-line with included CLI tools.
API First, CLI Enabled


Bulletproof Trust has been assessed and found awardable by the Chief Digital and Artificial Intelligence (CDAO) office and the Air Force Platform One Marketplace team.
Trusted by our Best Agencies
TRL9
Bulletproof Trust is Technology Readiness Level 9 (TRL9), deployed by government entities and commercial enterprises in SaaS, on-prem, private-cloud, and air-gapped environments.
Deployed and Battle Tested
If you are preparing a 510(k), PMA, De Novo, or HDE submission and need help with FDA cybersecurity documentation, medical device SBOM requirements, software supply chain risk management, vulnerability monitoring, or postmarket patch and update workflows, Bulletproof Trust can help your team move faster with stronger evidence and less manual effort.

Bulletproof Trust integrates with artificial intelligence large language models (AI LLM) through the Model Context Protocol (MCP). Get answers and deep insights on your risk data.
AI LLM Integrated

Bulletproof Trust is a 100% US owned and operated company developed by 100% US citizens, cleared or clearable.
Made in the USA

No complex calculations. No API call or user tracking. Our pricing is transparent, simple to understand, scalable across the smallest projects to the largest enterprises.
Transparent Pricing